Food Histamine Metabolism
Histamine, a molecule that exists in all foods, whether of animal or vegetable origin, must also be metabolized in order to be eliminated in the urine without inconvenience. It should be noted that exogenous histamine has no functional role in the body and is therefore eliminated without benefiting from any properties.
There are two major routes of histamine metabolism in living organisms, involving the enzymes Histamine-N-Methyltransferase (HMT) and Diamine Oxidase (DAO).
DAO is the most important enzyme in histamine metabolism, but it is only found in certain tissues: the intestinal mucosa, kidneys, placenta, thymus and seminal vesicles. It also plays a small role in liver tissue. It is mainly located in the intestinal epithelium, the area where histamine is absorbed and degraded by DAO, representing itself as a modulator of its passage into the bloodstream.
If you are interested in following a low histamine diet, we recommend you contact a healthcare professional specialized in DAO Deficiency.
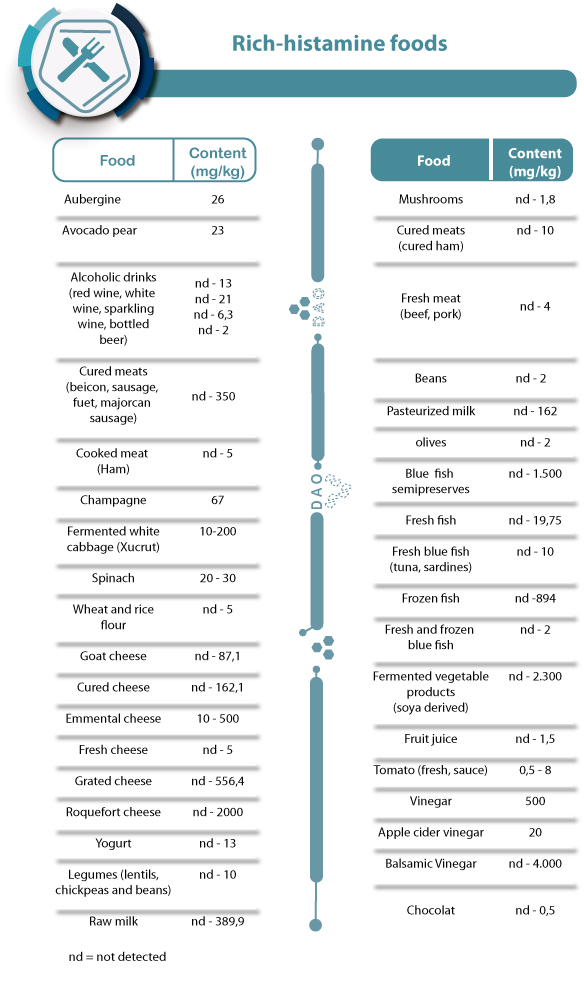
Table of rich-histamine foods (histamine content in mg/kg)























